Pure Astaxanthin Powder: What Should Buyers Look For?
When thinking about what buyers should look for in a high-quality astaxanthin extract, the main points are purity measurements that can be checked, clear explanations of the extraction process, pure astaxanthin powder and strict supplier certification. The best natural astaxanthin comes from Haematococcus pluvialis microalgae and has better stereoisomer profiles, especially the (3S, 3'S) configuration, which means it has a lot more bioactivity than synthetic petrochemical options. Professionals in charge of buying things should demand full analytical records, such as HPLC assay certificates showing concentration accuracy ranging from 1% to 96%, stability data under accelerated oxidation testing, and validation of microbial limits by a third party. In addition to the quality of the ingredients, the integrity of the production process is very important. For example, supercritical CO₂ extraction protects delicate molecular structures, and microencapsulation technology stops photodegradation while the product is being stored and mixed.
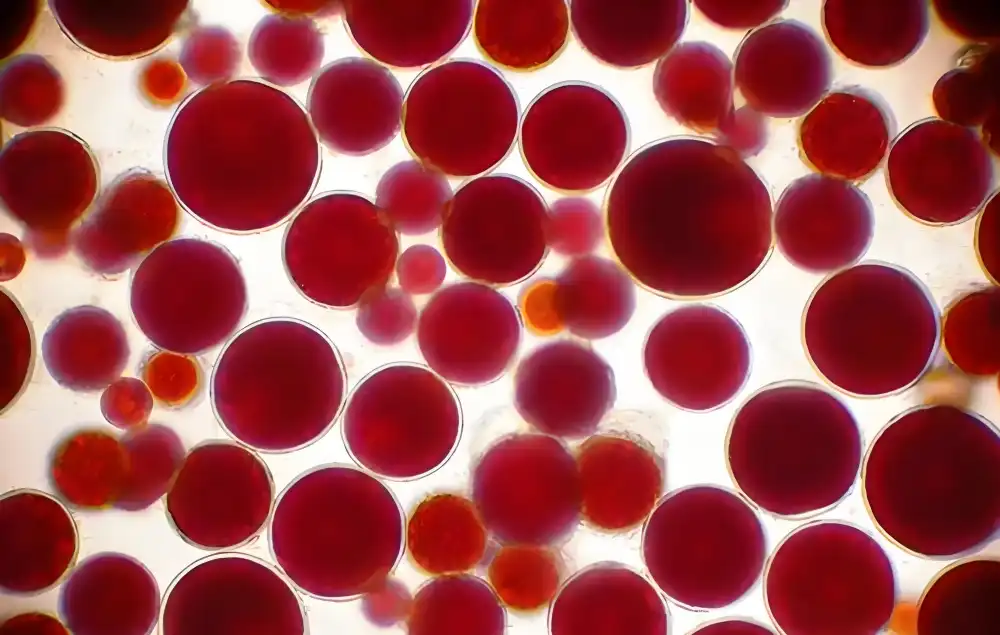
Understanding Pure Astaxanthin Powder: Composition, Benefits, and Uses
Origin and Molecular Characteristics
Natural astaxanthin is a keto-carotenoid colour that is made by Haematococcus pluvialis microalgae when they are stressed. The biologically better (3S, 3'S) stereoisomer comes from this plant source, which makes it fundamentally different from manufactured racemic mixtures made from petrochemical precursors. The chemical formula for C40H52O4 includes conjugated double bonds and strategically placed hydroxyl groups. This allows the compound to cross cellular membrane bilayers, which is a structural advantage that makes antioxidant activity easier in both lipophilic and hydrophilic cellular environments. It usually appears as a brown-red to dark violet powder, and its CAS registry number 472-61-7 confirms its chemical identity. The concentration ranges for high-quality materials from 1% to 96%, with pharmaceutical-grade products being at the upper end of this range. Advanced analytical methods, like HPLC and UV spectrophotometry, give accurate measurements, making sure that each batch is the same, which is very important for business formulation work. The extraction process has a big effect on the integrity of the finished product. For example, supercritical CO₂ fluid extraction technology keeps the bioactivity without leaving behind any solvent residues, while less effective methods may add contaminants or break down delicate molecular structures.
Health Benefits and Functional Applications
Scientists have found that astaxanthin is an extremely powerful antioxidant. In singlet oxygen quenching tests, it is about 6,000 times stronger than ascorbic acid and 550 times stronger than alpha-tocopherol. Multiple therapeutic uses can be found in a wide range of business sectors thanks to this amazing ability to neutralise free radicals. A main area of application for astaxanthin is skin health. It protects against UV-induced photoaging, reduces fine lines, and improves moisture retention through processes that keep collagen healthy. Clinical studies show that consistent supplementation improves the elasticity and texture of the skin. Eye health is another important benefit, as research has shown that astaxanthin crosses the blood-retinal barrier to protect photoreceptor cells from oxidative damage. Because of this, the substance is useful for eye health products that help with digital eye strain, age-related macular problems, and improving vision. Astaxanthin can help keep lipid levels healthy and control inflammatory pathways, which is useful for cardiovascular and metabolic health. The compound also protects neurons, which makes it useful for cognitive health supplements that aim to improve memory and mental clarity. For dietary supplements, the recommended daily dose is usually between 4 mg and 12 mg, but this can change depending on the health claims and the people who will be taking the supplements. The safety profile is still good—extensive toxicology studies confirm that the compound is safe at the suggested intake levels, and it is generally thought to be safe when it comes from natural extraction processes that are controlled for quality.
Industrial Applications Across Sectors
Astaxanthin is added to drug formulations and active pharmaceutical ingredients by pharmaceutical companies because it has anti-inflammatory and antioxidant qualities that help with therapeutic goals. The compound's ability to change membrane permeability to make other actives more bioavailable adds value to the mixture beyond its own benefits. The biggest market group is made up of nutraceutical and dietary supplement companies, which put the ingredient in capsules, tablets, softgels, gummies, and powder. Astaxanthin is very flexible and can be delivered in several different ways, such as oil-based suspensions that work best in softgels or cold water-dispersible forms that can be used in beverages. High-end cosmetic and personal care brands use it in anti-aging serums, moisturisers, and sun protection products. In addition to its useful properties, the natural red-orange colour of the ingredient makes it look nice. Companies that make food and drinks use astaxanthin as a natural colourant that also has health benefits. This makes products that look good and are good for you. Consumers want clean-label ingredients that can do more than one thing, so this dual functioning makes sense. For developing analytical methods, bioavailability studies, and formulation optimisation work, research institutions and labs need small batches of high-purity reference standards. They also need suppliers who can provide precisely characterised material with full certificates of analysis.
Comparing Pure Astaxanthin Powder with Other Forms and Supplements
Powder Versus Capsule and Oil Formats
The powder format has clear buying benefits for companies that need to be able to change the recipe easily and quickly process large amounts of powder. Unlike pre-encapsulated goods, powder lets you precisely control the dose during production and makes it easy to mix with other ingredients that work well together. Powder can be processed with standard tools like tablet presses, capsule filling machines, and powder sachet packaging lines, which is good for businesses that make things. Powder forms, which can be as concentrated as 96%, help formulators reach their goal potency levels while minimising the amount of inactive ingredient. Capsule and softgel formats are convenient for end users but limit the manufacturer's options. These finished tablet forms have set amounts of ingredients and encapsulation materials, which makes it harder to make them fit your needs. For some uses, oil-based astaxanthin suspensions are better, especially when making softgels, because the oil makes the astaxanthin more bioavailable through lipid-mediated absorption routes. Leading suppliers use microencapsulated powder technology to bridge this gap. Beadlet formations protect astaxanthin molecules while allowing them to dissolve in cold water. This means that raw astaxanthin can now be used in beverage systems and aqueous formulations that weren't possible to use before. Bioavailability factors have a big impact on format choice. Because astaxanthin is lipophilic, it needs the right delivery systems to make it easier for the body to absorb it in the intestines. When made correctly, powder goods that use advanced microencapsulation with food-grade carriers have bioavailability levels that are similar to oil formulations. They also have better stability profiles during storage and distribution. When it comes to medium- to large-scale production, cost-benefit analysis usually favours powder procurement, since bulk price and formulation flexibility make up for the need for extra processing steps.
Natural Versus Synthetic Astaxanthin Comparison
Astaxanthin that comes from algae and astaxanthin that is made in a lab are not only different in terms of where they come from, but also in terms of their stereochemistry, biological activity, and regulatory placement. The natural astaxanthin from Haematococcus pluvialis mostly has the (3S, 3'S) stereoisomer, which is the same molecular structure as pure astaxanthin powder found in marine animals at all levels of the food chain. This isomer has a lot more antioxidant power and cellular uptake efficiency than the racemic mixture (3S,3'S: 3R,3'S: 3R,3'R in a 1:2:1 ratio) that is typical of synthetic production. Regulatory frameworks are favouring natural sources more and more, with organic certification, non-GMO verification, and clean-label positioning only possible through botanical extraction pathways. Natural ingredients are becoming more and more popular among consumers, especially in high-end markets where product differentiation is key to buying choices. Many places don't allow synthetic astaxanthin to be used in human nutrition. Instead, it is mostly used in fish feed and not in nutraceutical or cosmetic products. Quality-conscious brands know that natural astaxanthin's higher price is justified by its better marketing, stronger claims of effectiveness, and wider acceptance by regulators. Another important factor that sets one brand apart is how openly the product is made. Controlled growth of microalgae and then tested extraction methods are used to make natural astaxanthin. This creates supply chains that can be tracked and quality control records. Chemical synthesis from petroleum derivatives is used in synthetic routes, which raises worries about residual solvents, reaction intermediates, and the long-term health of the environment. Brands that want to build a real wellness image and environmentally responsible sourcing narratives need to get their natural astaxanthin from GMP-certified facilities.
Comparison with Other Antioxidant Ingredients
Astaxanthin is a special kind of antioxidant that has benefits that make it stand out from other antioxidants like beta-carotene, lycopene, lutein, and choices that come from the sea, like krill oil. Beta-carotene and other carotenoids are good for you, but astaxanthin's structure, which has both hydroxyl and ketone functional groups, makes it better at integrating into membranes and protecting more cells. This difference in structure leads to significantly higher ORAC values and more complete free radical neutralisation in a wide range of oxidative stress situations. Krill oil contains astaxanthin and omega-3 fatty acids, which makes combination products appealing for some formulas. Finding a specific source of astaxanthin powder gives makers more control over the formulation, letting them hit exact potency goals without limiting fatty acid profiles or taking into account marine allergens. Microalgae-derived powder has a much higher astaxanthin content per unit volume than krill-based sources because it is more concentrated. This makes manufacturing more efficient and gives you more options for dosing the end product. Pure astaxanthin powder is also better for stability profiles because advanced microencapsulation technology protects it better against oxidation than oil-matrix delivery systems that can go bad. When choosing ingredients for a product, the procurement strategy should be in line with its positioning and formulation goals. Astaxanthin is great for uses that need the strongest antioxidants, claims about skin health, eye health support, and top market positioning. The scientific proof of the ingredient from a lot of clinical research backs up marketing promises with strong evidence, setting formulations apart in competitive market segments. More and more, buyers who want to improve the performance of formulations while still keeping clean label credentials choose natural astaxanthin powder as their first choice for a raw material.

Key Criteria When Choosing Pure Astaxanthin Powder for Procurement
Purity Standards and Analytical Verification
As part of the procurement requirements, full analytical documentation should be required, along with purity standards set by tested and proven methods. HPLC analysis is the best way to measure how much astaxanthin there is because it gives exact concentration readings, can tell the difference between stereoisomers, and finds possible breakdown products. Full spectral data, comparisons of retention times to trusted reference standards, and measurements of all-E versus Z-isomer ratios should all be included in certificates of analysis. UV spectrophotometry is another way to check the quality of a substance. It is especially useful for quickly screening batches and making sure the quality stays the same throughout production runs. Materials can be specified from 1% to 96% purity, with pharmaceutical and research-grade materials usually being purer than 95%. It is very important to understand the connection between the stated concentration and the carrier matrix composition. Lower percentage formulations use food-grade excipients that make them more stable and easier to process, while ultra-high purity grades are used in specific situations that need very few inactive ingredients. Heavy metal testing should show that amounts are less than 10ppm, with a focus on mercury, arsenic, lead, and cadmium. Microbial contamination limits must meet the standards needed for the application, and total plate counts, yeast, mould, and pathogen screening must be recorded for each production lot. Buyers should ask for accelerated stability data showing that the product stays effective at controlled temperatures and humidity levels. This knowledge predicts how long something will last and tells you how to store it properly. Particle size distribution analysis is important, especially for powder uses that need specific profiles for flow or dissolution. Limiting the amount of moisture to less than 5% stops hydrolytic breakdown and keeps the powder's ability to move freely during handling and processing.
Certifications and Regulatory Compliance
GMP certification is a basic requirement that shows that the quality control in manufacturing is in line with pharmaceutical standards. Facilities should show that they are currently certified by reputable organisations and make audit records available for buyers to look over upon request. With ISO standards like ISO 9001 for quality management systems and ISO 22000 for food safety management, you can be even more sure that your operations are running smoothly and that your processes are always the same. Organic certification from the USDA, EU, or similar organisations lets natural products stand out and appeals to health-conscious consumers who care about the long-term health of agriculture. Non-GMO certification meets the growing demand for genetic engineering transparency in the market, which is especially important given the ongoing debates about how to grow microalgae. Third-party testing by independent labs adds credibility above and beyond self-certification by the seller. Validation from organisations like NSF International, Informed Choice, or USP is recognised. Allergen-free claims prove the absence of common substances that cause reactions, making formulations more compatible across a wider range of product categories. Halal and Kosher certifications open up the market to certain religious groups, which increases the number of possible distribution channels. Regulatory compliance paperwork should include detailed dossiers that support import requirements across all target markets. Getting registered with the FDA to sell your product in the U.S., getting Novel Food approval to sell your product in Europe, and getting country-specific licenses for other places is what most people expect. Proactive suppliers who provide regulatory information and documentation support add a lot of value by cutting down on import delays and compliance uncertainty. Traceability protocols keep track of how materials move from growing microalgae to extracting, processing, packaging, and distributing them. This makes supply chains clear, which is important for meeting due diligence requirements and conducting quality incident reviews.
Supplier Evaluation and Quality Control Systems
To figure out how reliable a supplier is, you have to look at more than just the product specs. Some signs of a factory's ability to meet steady demand without lowering quality are the number of tanks used for extraction, the level of automation in the production line, and the number of batches that can be produced. Facilities that run 14 stainless steel extraction tanks with sizes between 500 and 3,000 litres and two fully automatic production lines show that they have the infrastructure to meet regular needs for large amounts of goods. 98% production efficiency means that processes are running at their most efficient, reducing waste and keeping the company competitive. Technical skills, such as having HPLC systems, UV spectrophotometers, rotary evaporators, and other related equipment in-house, allow for real-time quality monitoring and quick responses to questions about specifications. Dedicated research and development (R&D) teams show a dedication to continuous growth and the ability to create custom formulations that meet the specific needs of buyers. Quality control plans should include checking the raw materials, keeping an eye on the work as it's being done, testing the finished product, and checking for stability. Reliable quality assurance is based on well-documented standard operating procedures, trained staff, and properly calibrated equipment. The speed and technical support skills of suppliers have a big effect on the success of long-term partnerships. In addition to fulfilling orders, partners who offer formulation advice, regulation guidance, and troubleshooting help are good for buyers. Customer reviews, case studies, pure astaxanthin powder, and industry examples can tell you a lot about how well a supplier does in the real world. When possible, site checks let you see firsthand how things are made, how well they are kept, and how openly operations are run. Setting up clear lines of communication with specialised account management and technical contacts makes it easier to solve problems and helps build relationships where people work together.
Pricing Dynamics and Packaging Considerations
Pricing structures for natural astaxanthin powder reflect multiple cost drivers, including raw material sourcing, extraction technology sophistication, quality control rigor, and market positioning. Bulk purchasing typically unlocks volume discounts, with price breaks occurring at quantities ranging from 25kg to 100kg and larger. Understanding total landed cost requires evaluating not just unit pricing but also freight terms, customs duties, and payment conditions. Long-term supply agreements often secure more favorable pricing through commitment volumes while protecting against market volatility and raw material cost fluctuations. Packaging directly impacts product stability and handling efficiency throughout distribution and storage. Vacuum-sealed aluminum foil bags provide superior protection against light, oxygen, and moisture—the primary degradation vectors for astaxanthin molecules. Fiber drums with internal double-layer food-grade poly bags accommodate bulk quantities while maintaining material integrity during transportation and warehouse storage. Packaging should include clear lot identification, manufacturing and expiration dates, and handling instructions supporting proper storage protocols. Buyers managing inventory across multiple production facilities benefit from suppliers offering flexible packaging configurations matching specific operational requirements. Storage guidelines typically recommend refrigeration below 8°C in light-proof containers to maximize shelf life and preserve potency. Understanding supplier recommendations and incorporating appropriate cold chain logistics prevents quality degradation between receipt and use. Delivery timeframes ranging from one to three working days for standard orders indicate responsive supply chain management and adequate inventory positioning. Evaluating supplier lead times against internal production scheduling requirements ensures seamless material availability without excessive safety stock burdens or expedited shipping costs.
How to Source and Order Pure Astaxanthin Powder: Practical Tips for B2B Buyers
Strategic Sourcing Channels and Supplier Discovery
Identifying qualified suppliers requires strategic utilization of multiple discovery channels matched to specific procurement objectives. Specialized B2B marketplaces focusing on botanical extracts and nutraceutical ingredients provide concentrated supplier databases with search functionality enabling specification-based filtering. These platforms often include supplier verification systems, transaction protections, and comparative quotation tools, streamlining initial vendor identification. Industry trade exhibitions such as SupplySide West, Vitafoods Europe, and Fi Asia offer direct engagement opportunities with manufacturers, allowing face-to-face technical discussions, sample collection, and relationship building impossible through purely digital channels. Professional associations, including the Natural Products Association, Council for Responsible Nutrition, and regional industry groups, maintain supplier directories and facilitate networking among procurement professionals. Industry publications and technical journals provide market intelligence on emerging suppliers, technological innovations, and competitive landscape developments. Direct outreach to manufacturers operating in key production regions—particularly those with established microalgae cultivation infrastructure—uncovers suppliers potentially underrepresented in mainstream marketing channels but offering competitive advantages through vertical integration or specialized capabilities. Referrals from industry colleagues, contract manufacturers, and formulation consultants leverage collective experience identifying reliable partners. Requesting references from prospective suppliers and conducting due diligence through industry networks validates claims and reveals potential concerns before commitment. A multi-channel approach combining digital research, trade show participation, and professional networking maximizes supplier pool diversity while supporting comprehensive evaluation against procurement criteria.
Supplier Vetting and Due Diligence Protocols
Thorough supplier assessment begins with credential verification, examining certifications, regulatory registrations, and quality system documentation. Requesting copies of GMP certificates, ISO registrations, and third-party audit reports confirms claimed capabilities and reveals inspection history. Reviewing certificates of analysis from recent production batches establishes analytical methodology rigor and specification consistency. Comparing stated capabilities against observable infrastructure through virtual facility tours or promotional materials identifies discrepancies warranting clarification. Customer references provide critical insights into supplier performance across multiple operational dimensions. Speaking directly with existing clients reveals practical experience regarding delivery reliability, technical support responsiveness, quality consistency, and issue resolution approaches. Requesting case studies documenting successful partnerships illuminates supplier capabilities supporting complex requirements or challenging applications. Online reputation research through industry forums, review platforms, and business intelligence services uncovers potential concerns not disclosed during direct interactions. Sample evaluation represents an essential validation step before bulk commitments. Requesting representative samples matching the intended specification enables direct assessment of physical characteristics, analytical verification through independent testing, and preliminary formulation trials confirming compatibility with planned applications. Comparing samples from multiple suppliers under controlled conditions generates objective performance data informing final selection. Documenting sample evaluation results creates a reference baseline for ongoing quality monitoring following supplier selection.
Order Processing and Contract Negotiation
Clear purchase specifications documented in writing prevent misunderstandings and establish accountability frameworks. Detailed specifications should enumerate concentration requirements, analytical methods, acceptance criteria, packaging configurations, labeling requirements, and delivery schedules. Including quality agreements outlining testing protocols, dispute resolution procedures, and corrective action processes protects both parties through transparent expectations. Understanding minimum order quantities, standard lead times, and available shipping methods aligns procurement planning with supplier capabilities. Contract negotiation should address pricing structures, payment terms, volume commitments, and long-term supply agreements when appropriate. Discussing potential for supply interruptions and contingency protocols establishes risk mitigation strategies protecting production continuity. Clarifying intellectual property considerations, particularly for custom formulations or private label arrangements, prevents future conflicts. Including flexibility provisions addressing specification modifications, packaging changes, or delivery schedule adjustments accommodates evolving business needs without requiring complete contract renegotiation. Import compliance requires understanding destination country regulations governing natural ingredient importation, documentation requirements, and customs procedures. Suppliers experienced in international trade provide valuable guidance in navigating these complexities while supplying necessary documentation including commercial invoices, certificates of origin, phytosanitary certificates, and certificates of free sale. Establishing clear communication protocols with designated contacts for order management, technical questions, and urgent issues streamlines ongoing relationship management. Documenting all communications and maintaining organized procurement records supports quality system requirements and facilitates internal auditing.
Emerging Trends and Future Outlook of Pure Astaxanthin Powder in the Global Market
Technological Innovations in Extraction and Processing
Advanced extraction technologies continue evolving, improving yield efficiency, product purity, and environmental sustainability. Supercritical CO₂ extraction has largely displaced traditional solvent-based methods among quality-focused manufacturers, eliminating residual solvent concerns while preserving delicate molecular structures through controlled temperature and pressure parameters. Emerging refinements include sequential extraction protocols targeting specific lipid fractions and optimized microencapsulation matrices enhancing stability and bioavailability. Membrane separation technologies enable precise fractionation and purification, achieving pharmaceutical-grade purity levels with reduced processing steps and improved cost efficiency. Microalgae cultivation innovations focus on optimizing growth conditions, strain selection, and harvest timing to maximize astaxanthin biosynthesis. Photobioreactor designs incorporating LED lighting systems tuned to specific wavelength spectra enhance productivity while reducing energy consumption. Genetic characterization without modification identifies naturally high-producing strains, supporting non-GMO positioning while improving commercial viability. Integrated biorefinery concepts extract multiple value streams from microalgae biomass, reducing waste and improving overall process economics through comprehensive feedstock utilization. Analytical method development continues advancing, with rapid screening technologies enabling real-time quality monitoring during production. Portable spectroscopic instruments bring laboratory-quality analysis to production floors, facilitating immediate process adjustments and maintaining specification targets. Enhanced stability testing protocols predict shelf life performance more accurately, guiding packaging selection and storage recommendations. Digital quality systems incorporating blockchain technology create immutable traceability records, supporting supply chain transparency demands from regulators and conscious consumers.
Market Growth Drivers and Application Expansion
As the global wellness trend toward preventive healthcare and natural ingredients grows, so does the demand for astaxanthin in many different industries. As people become more aware of the health benefits of antioxidants, the supplement market grows. Astaxanthin is being used more and more in multivitamins, sports nutrition products, and supplements for specific health problems. Beauty-from-within ideas that link internal supplementation with external appearance outcomes speed up cosmeceutical uses, blurring the traditional category lines between nutrition and personal care. As populations age in developed markets, more people will be interested in products that help with age-related health issues like eye health, brain health, heart health, and skin ageing. As more clinical studies are done, more evidence is being gathered to support these uses. This lets marketers make stronger claims and doctors give more advice. New uses in sports performance and recovery take advantage of astaxanthin's ability to lower oxidative stress caused by exercise and speed up muscle recovery. Pure astaxanthin powder, which appeals to people who live active lifestyles. Concern for the environment affects buying decisions all along the supply chain, favouring natural astaxanthin derived from microalgae cultivation over synthetic alternatives that depend on petroleum feedstocks. Positioning products as plant-based or vegan is in line with changes in food preferences, especially among younger consumers. Natural astaxanthin is better than synthetic antioxidants and preservatives in food and drink because it has a clear label and doesn't have any added chemicals.
Strategic Considerations for Procurement Professionals
Successful procurement strategies predict how the market will change and put businesses in a good situation to take advantage of new opportunities and problems. Having ties with several qualified suppliers makes the supply chain more resilient against possible problems and gives you more negotiating power. Monitoring changes in regulations across target markets allows for proactive compliance positioning and saves the need for expensive re-formulation. Investing in partnerships with suppliers through technical cooperation and long-term commitments ensures priority allocation during times of tight supply. Differentiating premium products from commodity-level products is becoming more and more important, which is why investing in better raw materials that show clear performance benefits is a good idea. Sustainability claims, ethical sourcing, and environmental responsibility are all backed up by evidence that adds value to a brand beyond its useful benefits. Engaging suppliers with strong R&D capabilities helps with product innovation and formulation optimisation, which speeds up the time it takes to bring new product ideas to market. Industry participation, trade publications, and supplier relationships are all good ways to get early information on new technologies, competitive activities, and emerging uses. Planning for possible supply problems, price changes, or changes in regulations allows for quick responses that keep business going. When procurement workers add strategic value to their organisations through market research and managing relationships with suppliers, they have an effect that goes beyond just buying things.
Conclusion
Selecting the right natural astaxanthin ingredient requires balancing multiple technical, commercial, and strategic considerations. Product purity verified through comprehensive HPLC analysis, GMP-certified manufacturing, and transparent supply chain traceability form the foundation of quality procurement. Supplier capabilities encompassing adequate production capacity, responsive technical support, and demonstrated regulatory expertise enable successful long-term partnerships. Understanding format advantages—particularly microencapsulated powder's versatility and stability—guides optimal specification selection for diverse application requirements. Market trends favoring natural, sustainable ingredients position quality astaxanthin suppliers as valuable strategic partners supporting brand differentiation and product innovation. Successful procurement integrates technical rigor with commercial pragmatism, creating supply relationships delivering consistent value while supporting business growth objectives in competitive global markets.
FAQ
1. What distinguishes natural astaxanthin powder from synthetic alternatives?
Natural astaxanthin extracted from Haematococcus pluvialis microalgae contains predominantly the (3S, 3'S) stereoisomer, demonstrating superior biological activity compared to synthetic racemic mixtures derived from petrochemicals. Natural sources enable organic certification, non-GMO verification, and clean-label positioning unavailable with synthetic variants. Regulatory frameworks increasingly restrict synthetic astaxanthin to aquaculture applications rather than human nutrition, making natural sources essential for nutraceutical and cosmetic formulations. Consumer preference overwhelmingly favors natural ingredients, particularly in premium wellness categories where authenticity drives purchasing decisions.
2. How should buyers verify astaxanthin powder quality before bulk purchasing?
Comprehensive certificates of analysis documenting HPLC purity determination, heavy metal screening, microbial testing, and moisture content establish quality baselines. Requesting representative samples enables independent analytical verification through third-party laboratories and preliminary formulation compatibility assessment. Reviewing supplier certifications, including GMP, ISO, and organic registrations, confirms manufacturing quality systems. Customer references from established relationships provide practical performance insights regarding consistency, reliability, and technical support. Conducting supplier audits when feasible offers direct observation of facilities, equipment, and operational practices supporting quality claims.
3. What concentration specifications are appropriate for different applications?
Pharmaceutical applications typically require ultra-high purity grades exceeding 95% concentration for precise dosing and regulatory compliance. Dietary supplement formulations commonly utilize 2% to 10% concentrations incorporating microencapsulation matrices providing stability and processing advantages. Cosmetic applications employ similar mid-range concentrations, balancing potency with formulation compatibility. Food and beverage products generally use lower concentrations sufficient for functional benefits and natural coloring while remaining economically viable. Research applications demand precisely characterized reference standards with documented purity, enabling reproducible experimental results.
Partner with Fairir for Premium Natural Astaxanthin Powder Supply
Quality-conscious buyers seeking a reliable astaxanthin powder manufacturer should consider Shaanxi Fairir Biotech Co., Ltd. as their strategic sourcing partner. Our GMP-certified 10,000-square-meter facility houses advanced supercritical CO₂ extraction systems and 14 stainless steel tanks ranging from 500 to 3,000 liters, supporting consistent production capacity meeting demanding procurement schedules. We offer specification flexibility from 1% to 96% concentration, validated through HPLC and UV analytical methods, with comprehensive documentation supporting global regulatory requirements. Our product portfolio features organic, non-GMO, allergen-free, pure astaxanthin powder and vegan-certified options packaged in protective vacuum aluminum foil bags or fiber drums, ensuring stability throughout distribution. Delivery within one to three working days, competitive bulk pricing, and custom formulation capabilities, including private labeling, provide the operational flexibility your business requires. Our experienced technical team offers responsive communication and formulation support, helping you navigate complex ingredient specifications and application challenges. Request your complimentary sample today by contacting sales@fairirbiotech.com to experience the quality difference that distinguishes Fairir as your trusted long-term partner in premium botanical extract supply.
References
1. Ambati, R.R., Phang, S.M., Ravi, S., and Aswathanarayana, R.G. (2014). Astaxanthin: Sources, Extraction, Stability, Biological Activities, and Its Commercial Applications—A Review. Marine Drugs, 12(1), 128-152.
2. Capelli, B., Bagchi, D., and Cysewski, G.R. (2013). Synthetic Astaxanthin is Significantly Inferior to Algal-Based Astaxanthin as an Antioxidant and May Not Be Suitable as a Human Nutraceutical Supplement. Nutrafoods, 12(4), 145-152.
3. Higuera-Ciapara, I., Félix-Valenzuela, L. y Goycoolea, F.M. (2006). Astaxanthin: A Review of its Chemistry and Applications. Critical Reviews in Food Science and Nutrition, 46(2), 185-196.
4. Shah, M.R., Liang, Y., Cheng, J.J., and Daroch, M. (2016). Astaxanthin-Producing Green Microalga Haematococcus pluvialis: From Single Cell to High Value Commercial Products. Frontiers in Plant Science, 7, Article 531.
5. Naguib, Y.M. (2000). Antioxidant Activities of Astaxanthin and Related Carotenoids. Journal of Agricultural and Food Chemistry, 48(4), 1150-1154.
6. Guerin, M., Huntley, M.E., and Olaizola, M. (2003). Haematococcus Astaxanthin: Applications for Human Health and Nutrition. Trends in Biotechnology, 21(5), 210-216.










_1751965378790.webp)